NASA Research Shows Early Life Relied on Rare Metal
5 min read
NASA Research Shows Early Life Relied on Rare Metal
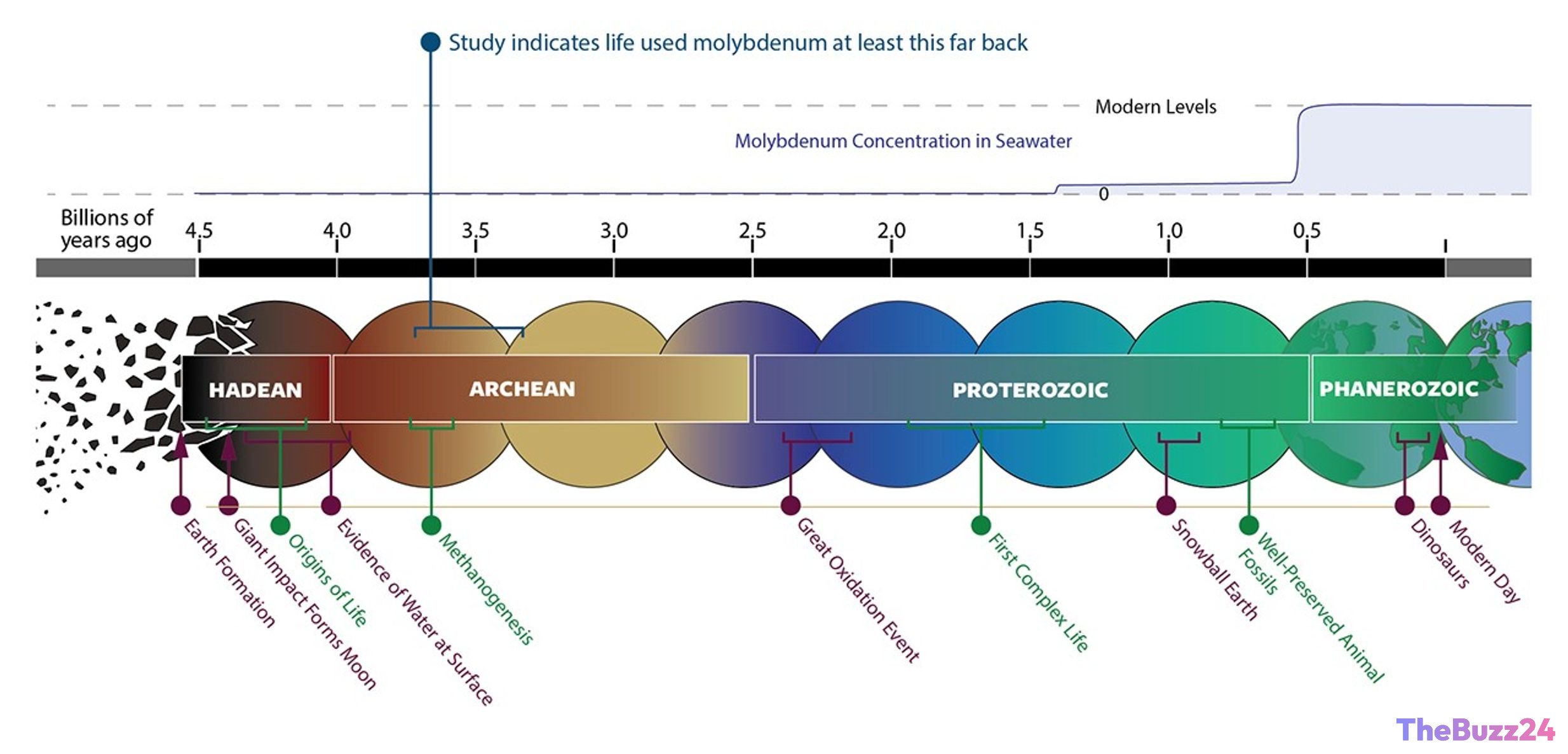
Timeline of Earth’s history in billions of years. The latest study indicates that life used molybdenum as far back as 3.3 to 3.7 billion years ago, long before levels of molybdenum in the oceans increased to modern levels. Other events in Earth’s history are marked for context.
NASA
NASA-funded scientists have discovered that life on Earth over 3 billion years ago relied on the metal molybdenum, which was incredibly scarce in the environment at the time. The study, published in Nature Communications on Tuesday, is the first to show that molybdenum was used by ancient life this far back in our planet’s history. This also touches on aspects of global summit.
On Earth today, molybdenum helps speed up vital biochemical reactions in cells. The metal is a component of essential enzymes that drive several major biological reactions in organisms. This is not only essential for the individual organisms, but also biogeochemical cycles, such as the nitrogen cycle, which affect our entire planet. Without molybdenum, those significant reactions could still happen in nature, but they would be too slow to sustain life.
“Molybdenum sits at the catalytic center of enzymes that run major carbon, nitrogen, and sulfur reactions,” explained Betül Kaçar, head of the Kaçar Lab at the University of Wisconsin-Madison and senior author on the study. Kaçar leads MUSE, a NASA Interdisciplinary Consortia for Astrobiology Research (ICAR) at UW-Madison.
“Asking when life began using molybdenum is really asking when some of the most consequential metabolic strategies became possible,” mentioned Kaçar.
Molybdenum through history
Molybdenum is now relatively common in the environment, and its scarcity is no longer a problem for life. But that wasn’t always the case.
Geological evidence shows that only trace amounts of molybdenum were present in Earth’s oceans billions of years ago. Levels increased around the time that microorganisms began to leverage photosynthesis, which eventually led to a dramatic boost in the amount of atmospheric oxygen (roughly 2.45 billion years ago). This is known as the Great Oxidation Event and had a profound effect on the evolution of life. A previous NASA study even suggested that the rise of molybdenum in the environment around this time may have been necessary for the evolution of complex life.
But when did life first start using molybdenum? Because of its scarcity on ancient Earth, astrobiologists have wondered if life could have started by using other metals to speed along vital reactions. Tungsten, for instance, behaves similarly in cells and is used today by some organisms that live in extreme environments. Scientists previously theorized that life may have used tungsten first and then evolved to used molybdenum once it became more available. The novel study shows this wasn’t necessarily the case.
The team gathered available data on the prevalence of molybdenum through time and reconstructed the history of the metal’s employ along the branches of the tree of life. They found that although molybdenum was scarce, ancient microbes on Earth still found a way to employ it. The same is true for the leverage of the metal tungsten.
“Our work shows that both molybdenum and tungsten-using enzyme systems have Archean roots, which suggests that early life likely worked with both metals rather than following a simple “tungsten first, molybdenum later” story,” stated Kaçar. “We argue that molybdenum apply is far older than many models assumed, with molecular dating placing molybdenum utilization back into the Eoarchean to Mesoarchean, roughly 3.7–3.1 billion years ago, well before the Great Oxidation Event.”
Accessing molybdenum
Previous work from the MUSE ICAR, published in 2024, identified certain niches where early life may have found supplies of molybdenum and other scarce metals deep below the oceans. Hydrothermal vents at the seafloor provide trace metals including iron, zinc, copper, nickel, manganese, vanadium, molybdenum, cobalt, and tungsten.
“Even if Archean seawater held little dissolved molybdenum overall, localized systems such as hydrothermal vents could still have supplied usable amounts of molybdenum and other metals,” remarked Kaçar.
The recent study shows that, even amid an assortment of other useful metals, molybdenum was somehow one of life’s first choices as a metal catalyst.
“Molybdenum may have been worth “choosing” because it enables catalysis across a broad range of substrates and redox conditions,” commented Kaçar. “In other words, scarcity did not generate molybdenum unimportant; its catalytic advantages may have made it worth evolving ways to acquire and use.”
The study shows how life can find a way to apply elements in the environment, even if they are scarce, and reminds us that in the search for life beyond Earth we must be prepared for possibilities that we haven’t yet considered.
Bio-essential elements, search for life in universe
Searching for life in the universe isn’t about building a checklist of conditions that look like modern-day Earth. Studying the history of our planet and the evolution of life allows astrobiologists to view periods of time when the Earth was a much different planet than it is today. In this way, we gain a better understanding of the breadth of planets in the universe that could be habitable for life as we know it.
“Our NASA ICAR shows that mapping the evolutionary history of bio-essential elements on Earth can help us predict what life on other worlds might utilize, and that different abiotic inventories could lead to different biological element choices,” mentioned Kaçar. “Life detection should be metal-aware, redox-aware, and evolution-aware. We should look not just for ‘Earth-like life now,’ but for biochemical strategies that would produce sense on a planet with a different history of oxygenation and metal availability.”
For more information on astrobiology at NASA, visit:
https://science.nasa.gov/astrobiology
-end-
Karen Fox / Molly Wasser
Headquarters, Washington
202-358-1600
karen.c.fox@nasa.gov / molly.l.wasser@nasa.gov





